From October 23-26, 2024, the Biomedical Engineering Society hosted their national meeting in Baltimore, MD. Many of UCLA’s Bioengineering department members attended to present their research, compete in student competitions, and participate in networking opportunities. This year UCLA was represented at the exhibit hall with faculty, staff, and student volunteers at the booth to advertise the department, recruit prospective candidates to our undergraduate and graduate programs, and promote research job opportunities. On October 24, 2023, the UC Systemwide Reception was held in the Hilton Key 1 & 2 Ballroom with all 10 UC campuses represented.
Below is the research presented and a slideshow of the weekend!
Undergraduate Students
Kelsey Fu, Undergraduate (2025)
Faculty Advisor: Dino Di Carlo
Research Title: Charge Asymmetry and Sequence Effects on Self-Coacervation of Intrinsically Disordered Proteins
Abstract: In aqueous environments, intrinsically disordered proteins (IDP) can often undergo liquid–liquid phase separation (LLPS), with the formation of a protein-rich liquid phase (coacervate) coexisted with a dilute supernatant phase. LLPS of IDP is a ubiquitous phenomenon underlying the formation of membraneless organelles that relates to myriads of essential cellular processes. The physiological function of the IDP coacervates is largely determined by their structure evolution and phase behavior. Significant progress has been made on the computational and experimental fronts in understanding the charge sequence effects on phase behavior, and structure-function relationship in self-coacervations of IDPs. However, most simulation studies assume that each protein has equal amount of positive and negative charges, while in nature typical IDPs always almost have some degree of charge asymmetry. It remains unclear how sequence effects under charge asymmetric conditions can regulate the thermodynamics and dynamics of self-coacervation in IDPs. This study explores the effects of varying sequence, charge asymmetry, and salt concentration on the self-coacervation of IDP using molecular dynamic (MD) simulations
Audrey Sogata, Undergraduate (2025)
Faculty Advisor: Daniel Kamei
Research Title: Developing a Quantitative Multicolor Paper-Based Device by Combining the Lateral-Flow Immunoassay with Gold Nanorod Etching
Abstract: Therapeutic drug monitoring is used to measure the concentrations of drugs, such as the cardiac medication digoxin, to ensure safe and effective dosages. Since many digoxin patients live in resource-limited areas, there is a need for an inexpensive, rapid diagnostic that can quantify this target molecule as the current gold standards require expensive equipment and trained personnel. The lateral-flow immunoassay (LFA) is a paper-based diagnostic but suffers from its inability to provide quantitative information. To address this, our lab previously combined LFAs with an H2O2 solution and gold nanorod (GNR) etching in suspension to yield easily distinguishable color hues that could be correlated to digoxin concentrations. However, this method required many liquid-handling steps that prevented its use at the point of care. This work expands upon our previous study to create a fully paper-based set-up combining LFAs with GNR etching on paper.
Lillian Gong, Undergraduate (2025)
Faculty Advisor: Daniel Kamei
Research Title:A Molecular Thermodynamic Model for Improving Point-of-Care Diagnostics Incorporating the Lateral-Flow Immunoassay and Aqueous Two-Phase Systems
Abstract: Although the lateral-flow immunoassay (LFA) has seen success as a point-of-care diagnostic, it suffers from low sensitivity when the target is present at low concentrations. To address this challenge, our group developed the Aqueous two-phase system (ATPS)-automated Concentration and Enhancement of the LFA (ACE-LFA), which utilizes ATPS separation on paper to both concentrate the sample and automatically and sequentially deliver signal enhancement reagents. This approach relies on the selective partitioning of 3,3’,5,5’-tetramethylbenzidine (TMB) and the target bacteria to opposite phases of the ATPS, which minimizes premature signal enhancement and ensures the delivery of the TMB enhancement reagent to the detection zone only after the bacteria has bound to the test line. Accordingly, the optimal combination of enhancement reagent and ATPS is one in which the enhancement reagent partitions significantly into the polymer-rich phase of a polymer-salt ATPS. Therefore, we developed a molecular thermodynamic model to predict this partitioning behavior. Using this model, we predicted the partitioning behavior of TMB and 3,3’-diaminobenzidine (DAB), another enhancement substrate, in a polymer-salt ATPSs and compared our predictions with experiments. We then confirmed our hypothesis that improved enhancement with darker test lines and minimal premature signal enhancement would be achieved when the enhancement reagent partitions extremely into the polymer-rich phase.
Cara Susilo, Undergraduate (2026)
Faculty Advisor: Mireille Kamariza
Research Title: Quantifying drug effects on Mycobacteria via fluorescence on a plate reader towards development of drug susceptibility test
Abstract: Tuberculosis (TB), caused by Mycobacterium tuberculosis (Mtb), is the second-most infectious disease estimated to infect more than 10 million people a year. About 4.5% of new cases report drug-resistance to one or more of the four first-line drugs, isoniazid (INH), rifampicin, pyrazinamide, and ethambutol. Increased resistance is attributed to inadequate medical systems and incorrect treatments informed by phenotypic susceptibility tests through directly observed therapy. Insufficient lab capacity, resources, and repeated courses of treatment can amplify resistance patterns in the local populations. The rapid transmission of TB in these communities highlights a need for a faster, more accessible, and reliable diagnostic. Current methods include the TB skin test, blood test, chest radiography, sputum smear microscopy, and interferon gamma release assays. Rapid molecular diagnostic tests recommended by the World Health Organization include the Xpert MTB/RIF Ultra and Truenat assays, which can detect drug resistance early; however, detection of multi-drug resistant TB, latent TB, and HIV-associated TB remains expensive, complex, and difficult for high TB burden communities to access. Several trehalose-based probes have been developed utilizing metabolic pathways that uptake trehalose for lipidation and insertion into the mycomembrane, a phenotype unique to mycobacteria. Trehalose conjugated to a solvatochromic fluorophore results in the mycobacteria-specific probe 4-N,N-Dimethylamino-1,8-naphthalimide (DMN-Tre), which can report drug susceptibility to INH in Mycobacterium smegmatis (Msmeg), a non-pathogenic close relative to Mtb.
Amelia Rodolf, Undergraduate (2026)
Faculty Advisor: Mireille Kamariza
Research Title: Solvatochromic trehalose probes for detection of dormant mycobacteria
Abstract: Tuberculosis (TB) remains the most deadly infectious disease—briefly surpassed by the COVID-19 pandemic—and heavily impacts low-income countries with scarce resources for detection and treatment. Approximately 1.7 billion people harbor latent TB infections, caused by Mycobacterium tuberculosis (Mtb) in a dormant, non-replicative state. These latent infections can progress to active, life-threatening TB at any time. The global eradication of TB necessitates innovative tools for point-of-care detection of dormant Mtb. Current diagnostic methods, such as sputum smear microscopy with Ziehl-Neelsen or auramine staining, often fail to detect dormant mycobacteria due to reduced acid-fastness during dormancy, resulting in false negative results. To address this challenge, we previously reported on a novel solvatochromic probe, 4-N,N-dimethylamino-1,8-naphthalimide-conjugated trehalose (DMN-Tre), which fluoresces upon metabolic incorporation into the mycomembrane of live Mtb, catalyzed by antigen 85 (Ag85). Here, we demonstrate the efficacy of DMN-Tre in labeling both active and dormant mycobacteria, using low-carbon nutrient media to induce dormancy. We show that dormant bacteria remain viable and rapidly reactivate in nutrient-rich environments. DMN-Tre labeling intensity correlates with metabolic activity, making it a promising probe for investigating metabolic processes. DMN-Tre offers a low-cost, rapid, and specific method for detecting dormant Mtb and shows potential both as a clinical diagnostic for latent TB and as a scientific tool for understanding mycobacterial metabolism.
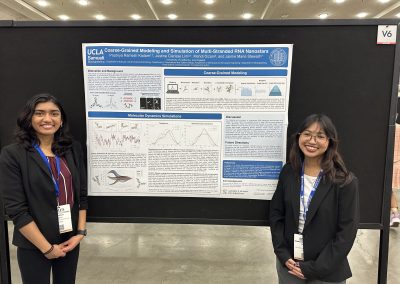
Pradnya Kadam, Undergraduate (2026)
Faculty Advisor: Jaimie Stewart
Research Title: Coarse-Grained Modeling and Simulation of Multi-Stranded RNA Nanostars
Abstract: Biomolecular condensates are membraneless organelles which are phase-separated compartments of RNA and protein. These biological compartments are responsible for the spatial and temporal organization of molecules and biochemical reactions. Our recent work has demonstrated the ability to design and synthesize artificial RNA condensates using multi-stranded RNA motifs consisting of three- and four-way junctions, called RNA nanostars. Nanostars bind to each other through programmed single-stranded overhangs, called sticky-ends. These artificial condensates have the potential to be utilized to study biological processes as well as design novel biosensors and drug delivery mechanisms. Studying the behavior of these structures will help us gain insight into developing useful artificial RNA condensate systems.
Justine Lim, Undergraduate (2026)
Faculty Advisor: Jaimie Stewart
Research Title: Coarse-Grained Modeling and Simulation of Multi-Stranded RNA Nanostars
Abstract: Biomolecular condensates are membraneless organelles which are phase-separated compartments of RNA and protein. These biological compartments are responsible for the spatial and temporal organization of molecules and biochemical reactions. Our recent work has demonstrated the ability to design and synthesize artificial RNA condensates using multi-stranded RNA motifs consisting of three- and four-way junctions, called RNA nanostars. Nanostars bind to each other through programmed single-stranded overhangs, called sticky-ends. These artificial condensates have the potential to be utilized to study biological processes as well as design novel biosensors and drug delivery mechanisms. Studying the behavior of these structures will help us gain insight into developing useful artificial RNA condensate systems.
Graduate Students
Karina Bender, MS (2025)
Faculty Advisor: Jeffrey Hsu
Research Title: Development of a microfabrication pipeline to pattern ECM proteins and guide EndMT mechanobiology
Abstract: Endothelial-to-mesenchymal transition, or EndMT, is the process where endothelial cells undergo molecular changes that shift their phenotype from that of endothelial cells to that of mesenchymal-like cells. This biological process is implicated in many physiological and pathological conditions, such as cancers and fibrotic diseases, and has been recognized as a mediator in atherosclerosis and vascular calcification, leading to research to determine whether the EndMT process is a suitable therapeutic target for the treatment of these pathologies. Studies in recent years have provided an improved understanding of how to assess EndMT; however, the marker expression signatures of EndMT are highly variable, and better methods are needed to characterize the EndMT continuum. It has also been shown that endothelial cells can undergo EndMT in response to mechanobiological forces, and while research has been done to control EndMT via proteins and culturing, little is known about the effect of micropatterns on EndMT mechanobiology and, by extension, its differentiation. Furthermore, studying the effects of micropatterns on any cell’s biology is both time and resource-intensive due to the costs of micropatterning, causing it to be out-of-reach for many research projects. We aimed to develop a novel and cost-effective protein-stamp fabrication, cell culture, and image analysis pipeline to study and control the EndMT process to aid in the identification of therapeutic targets for EndMT-related diseases, as well as for further applications in tissue engineering and regenerative medicine.
Rajesh Ghosh, Ph.D. (2024)
Faculty Advisor: Dino Di Carlo
Research Title: A Single-tier Multiplexed Lyme Disease Assay Enhanced by Machine Learning
Abstract: Lyme disease (LD) is the most prevalent vector-borne disease in North America and Europe (Fig 1a). Yet, the current two-tier centralized lab tests have significant drawbacks, including long turnaround times, underreporting of cases, often resulting in failure to detect and treat LD in its early stages when treatment is most effective at preventing widespread complications (Fig 1b). In addition, the tests used are either subjective or use epitopes that are limited in sensitivity and specificity leading to undesired outcomes. Rapid, multiplexed and low-cost diagnostic platforms that can assay the repertoire of antibodies formed against pathogens can improve the diagnosis of difficult to detect diseases such as Lyme disease, facilitating early detection and improving patient outcomes. In this work, we report a single-tier rapid detection of Lyme disease using a multiplexed vertical flow assay (xVFA) that employs synthetic peptides as diagnostic targets, complemented by machine learning-assisted disease classification (Fig 1c). Clinical testing from two US repositories, including the Bay Area Lyme Disease Biobank (LDB) and the CDC, shows that the single-tier xVFA matches the performance of standard two-tier testing (STTT). This indicates the potential to replace the cumbersome two-tier centralized lab test with a rapid, single-tier point-of-care assay.
Youcheng Yang, Ph.D. (2027)
Faculty Advisor: Song Li
Research Title: Synthetic Viscoelastic Activating Cells for T Cell Engineering and Cancer Therapy
Abstract: T cell-based therapies, particularly CAR-T cell therapy, hold great promise for cancer treatment. However, current methods for T cell activation and expansion, such as antibody-coated paramagnetic polystyrene beads (e.g., Dynabeads), often result in suboptimal cell subpopulations, low efficiency of CAR introduction into T cells, low stemness, and limited durability for long-lasting therapeutic effect. One reason is that Dynabeads are stiff and have a very different mechanical property in comparison to antigen-presenting cells (APCs) which are much softer and have the viscoelastic property that is critical to mediate ligand-receptor engagement and activation.
Barath Palanisamy, Ph.D. (2026)
Faculty Advisor: Dino Di Carlo
Research Title: Multiplexed Islet Autoantibody Detection via Vertical Flow Assay for Early Type 1 Diabetes Diagnosis
Abstract: Over 57 million people globally suffer from diabetes mellitus, predominantly in resource-limited areas. Diabetic ketoacidosis (DKA), a life-threatening condition resulting from insufficient insulin remains a major concern, causing over 50% of child fatalities from Type 1 Diabetes (T1D) and costing the US $5.1 billion annually. Early diagnosis of T1D can significantly reduce risks by enabling timely interventions to prevent or mitigate DKA (Fig. 1A). Islet autoantibodies (IAb) have been identified as critical predictive biomarkers where the presence of two or more of these autoantibodies—insulin (Ins), islet antigen-2 (IA-2), zinc transporter 8 (ZnT8), and glutamic acid decarboxylase (GAD65)—signals a 70% likelihood of developing T1D within ten years (Fig. 1B). Currently, no point-of-care (POC) test exists for simultaneous detection of these islet autoantibodies in a cost-effective and rapid manner for population-level screening. To address this gap, we created a Vertical Flow Assay (VFA) for multiplexed detection of T1D-associated autoantibodies through a rapid, low-cost test, delivering results in less than 20 minutes. Coupled with a smartphone reader for data acquisition and automated analysis, the VFA T1D test provides T1D risk classification and is well-suited for the early diagnosis of T1D in at-risk populations. (Fig. 1C).
Jing Wang, Ph.D. (2025)
Faculty Advisor: Tzung Hsiai
Research Title: Snai1b Mediates Myocardial Trabeculation and Growth in Response to Mechanical Stimulation
Abstract: Snail family genes have long been recognized as inducers of epithelial-to-mesenchymal transition (EMT) in various cell types involved in cardiac development and injury, including endocardial cells, epicardial cells, and fibroblasts. The activation of Snail genes is crucial to processes such as valvulogenesis, fibrosis, and regeneration. However, the activation of Snail genes within cardiomyocytes (CMs) has yet to be characterized.
Although recent works reported that a zebrafish Snail gene, snai1b, plays a role in guiding the migration of myocardial progenitors and maintaining the architecture of the cardiac wall, the spatiotemporal dynamics of snai1b expression and the subsequent modulation of trabeculation are still poorly understood.
Emily Bozich, Ph.D. (2026)
Faculty Advisor: Jennifer Wilson
Research Title: Protein interaction network topology predicts in vitro synergy comparable to approaches trained with experimental data
Abstract: Combination therapies are often regarded as the future for treating cancer. However, anticipating the synergistic effects that arise from combined perturbations (i.e., drug combinations) is challenging as it requires an intricate understanding of drug targets’ connections to a disease’s dysregulated molecular pathways. In fact, even sophisticated machine learning methods trained on ample single perturbation and cell line information have reported model-experiment Pearson’s correlation coefficients (PCC) of ~0.24-0.48, with better performing models leveraging drug target and pathway information. Further, network-based methods have successfully been applied to the protein-protein interaction network (interactome) in order to identify disease/signaling pathways, discover new drug targets, and even predict drug synergies. However, to our knowledge, few have considered de novo synergy prediction solely from interaction network topology.
Joyce Huang, Ph.D. (2025)
Faculty Advisor: Song Li
Research Title: Bio-ionic Hydrogel for Biodegradable, Ion-Conductive Peripheral Nerve Guides
Abstract:While peripheral nerves have some capacity for regeneration, the rate of regeneration is insufficient to restore function in many cases of peripheral nerve injury (PNI). Renervation following injury is impeded by axon apoptosis and muscle atrophy. Therefore, medical intervention is required to restore motor control in many cases of PNI. PNI are currently treated clinically using autografts. However, autografts are limited by the source and donor site morbidity. Biomaterial nerve conduits can be used to facilitate axon regrowth across large defects. Conductive biomaterials pose a particularly attractive option for fabrication of nerve guides due to their ability to conduct native electrical signals, accelerating axon extension. However, conductive materials commonly used for nerve conduit fabrication can be cytotoxic and have limited degradation in vivo. Using a biocompatible ionic liquid, we investigate the use of a biodegradable ion-conductive hydrogel in facilitating peripheral nerve regeneration.
Shaolei Wang, Ph.D. (2025)
Faculty Advisor: Tzung Hsiai
Research Title: A self-assembled implantable microtubular pacemaker for wireless cardiac electrotherapy
Abstract: The current cardiac pacemakers are battery-dependent, and the pacing leads are prone to introduce valve damage and infection, plus a complete pacemaker retrieval is needed for battery replacement. Despite the reported wireless bioelectronics to pace the epicardium, open-chest (thoracotomy) is required to implant the device, and the procedure is invasive, requiring prolonged wound healing and healthcare burden. We hereby demonstrate a fully biocompatible wireless microelectronics with a self-assembled design that can be rolled into a light-weight microtubular pacemaker for intravascular implantation and pacing. The radiofrequency was employed to transfer energy to the microtubular pacemaker for electrical stimulation. We show that this pacemaker provides effective pacing to restore cardiac contraction from a non-beating heart, and have the capacity to perform overdrive pacing to augment blood circulation in an anesthetized pig model. Thus, this microtubular pacemaker paves the way for the minimally invasive implantation of leadless and battery-free microelectronics.
Brian Arianpour, Ph.D. (2026)
Faculty Advisor: Tzung Hsiai
Research Title: Flexible On-Skin Electrical Impedance Tomography for Early and Remote Detection of Fatty Liver Disease
Abstract: Obesity is a rising pandemic and more than two third of Americans are overweight. Obesity-related non-alcoholic fatty liver disease (NAFLD) is a silent killer. If undetected, NAFLD progresses to liver cirrhosis and cancer. Electrical Impedance Tomography (EIT) is an emerging imaging modality for assessing tissue-specific electrical conductivity. Fatty infiltrate in the liver is characterized by its frequency-dependent electrical impedance (Z) in response to applied alternating current (AC). However, existing EIT systems are bulky and impractical for point-of-care or remote detection.